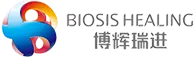As the Year of the Horse begins, Biosis Healing is pleased to announce a major regulatory milestone: its flagship Dural Repair Patch has been granted Class IV registration under Hong Kong’s Medical Device Administrative Control System (MDACS) and officially listed on the medical device register of the Department of Health of the Hong Kong Special Administrative Region.
This approval validates the company’s technological excellence, product quality, and end-to-end compliance system in the high-end implantable biomaterials sector. It provides full market access to Hong Kong and lays a solid foundation for Biosis Healing to expand into broader international markets and advance its global strategy.

As a core product from Biosis Healing’s proprietary SIS biomaterial platform, the Dural Repair Patch received China NMPA Class III medical device certification in 2021 and has since been widely adopted in clinical practice, delivering safe and reliable care for patients.
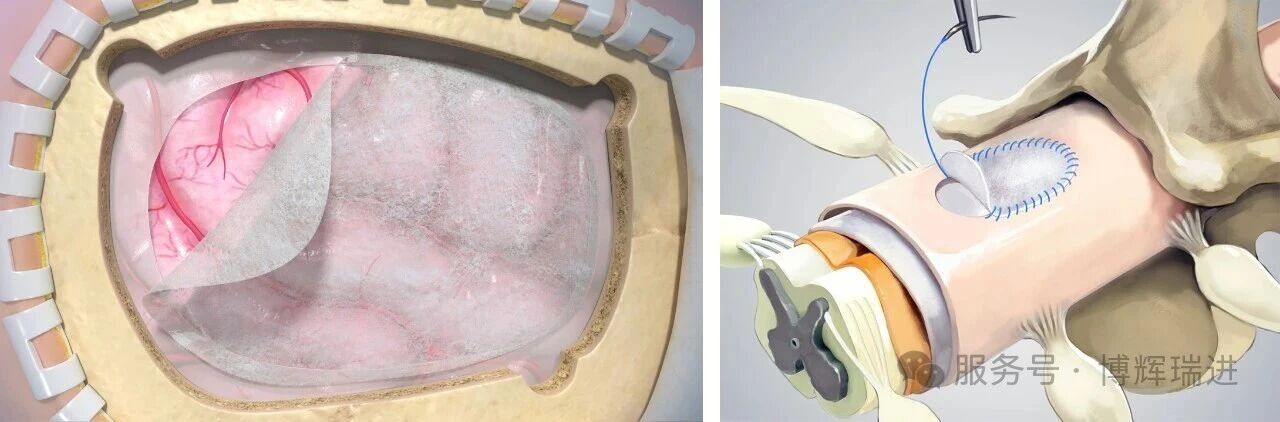
Suitable for a wide range of neurosurgical procedures, the product effectively lowers the risk of cerebrospinal fluid leakage. It offers ideal mechanical strength to support precise clinical procedures. Enriched with natural bioactive factors, the material induces endogenous tissue regeneration, degrades gradually and completely as new tissue forms, and demonstrates excellent anti-infective properties—making it a preferred alternative for dural repair.
Hong Kong is a leading hub for medical innovation and international medical technology exchange in Asia, with stringent MDACS regulatory standards. Class IV designation represents the highest risk category for medical devices, requiring comprehensive verification of safety, performance, and strict quality system audits—reflecting the product’s technical maturity and clinical reliability.
Biosis Healing’s successful approval underscores its innovative capabilities in advanced implantable biomaterials and the product’s value in protecting patient safety and enabling precise repair in complex surgical settings.
This Hong Kong market entry marks another key milestone in Biosis Healing’s global expansion. To date, the company has obtained regulatory clearances from major authorities worldwide, including China NMPA, EU CE, US FDA, and Vietnam MOH, establishing a sales network covering key global markets.
With the Hong Kong MDACS listing, Biosis Healing will be eligible for procurement by major public hospitals in Hong Kong, bringing high-quality domestic implantable medical devices to local patients.
Looking ahead, the company will actively pursue industry-university-research-hospital collaborations with Hong Kong medical institutions, universities, and research platforms to accelerate clinical adoption of the Dural Repair Patch in the region. Leveraging Hong Kong as an international gateway, Biosis Healing will strengthen global medical partnerships and expand its international footprint, continuing to deliver advanced solutions to patients worldwide.
Guided by its mission, the company remains patient-centric, using cutting-edge regenerative biomaterial technologies to provide comprehensive solutions for advanced surgical care.