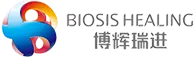Recently, Biosis Healing’s Class III medical device Dural repair patch has successfully passed review by the Ministry of Health of Vietnam and been granted the Class D Medical Device Registration Certificate. This official approval authorizes full market access, commercial sales, and clinical application in Vietnam. It marks a key step in expanding Biosis Healing’s footprint in Southeast Asia and sets a valuable example for China’s high-end implantable medical devices going global.

Vietnam classifies medical devices into four risk categories (Class A to D), with Class D being the highest-risk tier covering implantable devices and life-supporting equipment. Registration for Class D products requires strict full-cycle evaluation by Vietnam’s Ministry of Health, including rigorous assessment of product safety, clinical efficacy, quality management systems, and manufacturing compliance.
The successful approval of Biosis Healing’s Dural repair patch confirms the product’s exceptional safety and clinical performance. It also demonstrates that Biosis Healing’s quality systems, production standards, and risk control fully meet international benchmarks.
As one of Biosis Healing’s core products, the Dural repair patch is manufactured using proprietary SIS biomaterial developed in-house. It offers multiple clinical advantages:
· Suitable for a wide range of neurosurgical procedures and effective in preventing cerebrospinal fluid leakage
· Balanced mechanical strength for ease of surgical handling
· Rich in bioactive factors to support endogenous tissue regeneration and full absorbable degradation
· Excellent infection resistance, making it an ideal dural repair substitute
This certification strengthens Biosis Healing’s global layout and reinforces its commitment to delivering safe, innovative biomaterial solutions for patients worldwide.
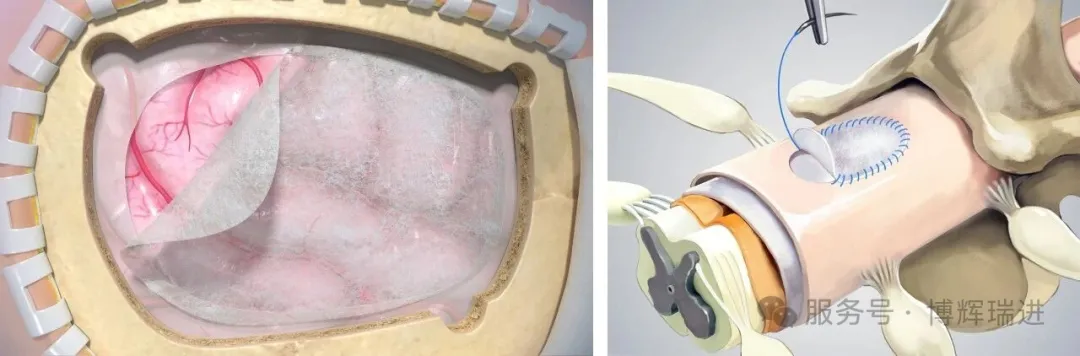
Image: Clinical Application Diagram of the Dural Repair Patch